Intranasal Preparations
Upon completion of this exercise, you should be able to:
- Describe the major physiological function of the nose.
- Describe four characteristics of an acceptable intranasal solution.
- List four intranasal preparations.
- Explain how to calibrate a dropper, a nasal spray bottle, or a bottle with an MDI pump.
- Counsel a patient about the proper administration of intranasal drops, sprays, or MDIs.
Physiology of the Nose
The nose is a facial structure made of cartilage and bones, and covered with skin. Its primary function is to serve as a channel for transporting air to the lungs as part of the respiratory system. The adult nasal cavity has a capacity of about 20 mL, a very large surface area for absorption, and a very rich blood supply. The air is warmed by the rich blood supply of the nose and humidified while passing through the nose on the way to the lungs. Dust, bacteria, and other matter are entrapped by mucus and swept by cilia through the nasal passageways until the materials can be swallowed, destroying much of the harmful material.
Nasal fluids are normally in the pH range of 5.5 to 6.5. Lacrimal fluids, as well as fluids from the four paranasal sinuses, drain through the nasal cavities, where they are also swallowed. The nose contains olfactory receptors that give the sense of smell which plays an important role in the complexity of tastes.
Intranasal Preparations
The common intranasal liquid dosage forms, solutions and suspensions, can be prepared as drops, squeeze sprays or bottles with MDI valves, nebules, and aerosol intranasal inhalers. Intranasal gels in water miscible base, which are applied to the inside of the nares, can also be prepared. Intranasal solutions should be isotonic, equivalent to 0.9% sodium chloride. The isotonicity will prevent damage to the cilia; damaged cilia cease their natural movements. Osmotic pressures ranging from 200-600 mOsmol/L are acceptable for intranasal administration, although the isotonicity of resident nasal fluids is about 300 mOsmol/L.
Listed below are many of the acceptable characteristics for intranasal solutions. To achieve these characteristics, intranasal preparations contain many of the same excipients fouud in ophthalmic formulations. This commonality is found in vehicles, buffers, preservatives, tonicity adjusting agents, gelling agents, and antioxidants.
| Characteristics of an Acceptable Intranasal Solution |
|---|
| pH in the range of 5.5-7.5 (4-8 is acceptable) Mild buffer capacity Isotonic (0.6%-1.8% sodium chloride equivalent is acceptable) Does not modify the normal mucus viscosity (e.g., not too hypertonic or hypotonic) Does not interfere with normal ciliary motion Does not interfere with normal ionic composition of nasal secretions Compatible with active ingredients Stable Sterile Preserved |
Sprays, MDIs, Aerosols
 Intranasal inhalations are administered to achieve either local respiratory or systemic therapeutic benefits. Special devices, such as the plastic spray bottle or bottles with metered dose valves, are used to break up a solution preparation into small particles and deliver a spray.
Intranasal inhalations are administered to achieve either local respiratory or systemic therapeutic benefits. Special devices, such as the plastic spray bottle or bottles with metered dose valves, are used to break up a solution preparation into small particles and deliver a spray.
 Some active drugs are administered using pressurized intranasal aerosols (i.e., an intranasal aerosol inhaler). The advantages and disadvantages of the intranasal aerosols are the same as those of the inhaled aerosols administered through the mouth.
Some active drugs are administered using pressurized intranasal aerosols (i.e., an intranasal aerosol inhaler). The advantages and disadvantages of the intranasal aerosols are the same as those of the inhaled aerosols administered through the mouth.
 The one-way nasal pumps (i.e., MDIs in bottles) prevent drawback contamination of nasal fluids into the bottle after administration.
The one-way nasal pumps (i.e., MDIs in bottles) prevent drawback contamination of nasal fluids into the bottle after administration.
Nebules
Other drugs are packaged in nebules and administered through nebulizers as “inhalation therapy.” The nebulizers dispense the drug particles into the 0.5-5.0 µm range, which is the optimum particle size for inhalation administration. The particles in the optimum particle size range are carried out of the nebulizer in a mist; the larger particles do not exit the device, but fall back into the reservoir of medication to be re-nebulized.
Inhalers
If the drug is sufficiently volatile, it can be administered via a nasal inhaler. The inhaler is a capped cylindrical tube that contains fibrous material impregnated with a volatile drug. The patient removes the cap and the inhaler tip is placed just inside the nostril. As the patient inhales, air is pulled through the tube and the vaporized drug is pulled into the nasal cavity.
Loss of Drug During Administration
Even through there are several different dosing devices for intranasal administration, the drug can still be lost following administration by three mechanisms:
- Metabolism in the nasal mucosa. The nasal mucosa is an enzymatically active tissue; some drugs are significantly degraded when administered by this route.
- Mucus flow and ciliary movement. The normal physiology of the nasal cavity is to move mucus and inhaled contaminants up and away from the lungs and toward the orifice of the nose. Some drug loss will occur as the drug is swept outward by these processes.
- Swallowing. Intranasal administration can also cause the drug to be swallowed and in some cases enough drug will be swallowed to be equal to an oral dose. This may lead to a systemic effect from the drug even though it is intranasally administered.
Calibration of Dropper or Spray Bottle
Generally, intranasal solutions and suspensions are administered as drops; solutions can also be administered as a fine mist from a nasal spray bottle. Nasal sprays are preferred to drops because drops are more likely to drain into the back of the mouth and throat and be swallowed.
If drops or sprays are used, the quantity of drug administered in each drop or each spray should be calibrated.
To calibrate a dropper:
- drop the formulation into a small graduated cylinder (5 mL or 10 mL) using the dropper the patient will use
- count the number of drops required to dispense 3 ml of solution
- divide the number of drops by 3; this will give the number of drops per mL
- calculate the number of drops needed to dispense the volume of formulation in one dose
To calibrate a spray bottle:
- prime the spray bottle
- weigh the spray bottle when filled with the formulation
- deliver 10 sprays into a plastic bag
- re-weigh the spray bottle
- divide the difference in the two weights by 10; this will give an average volume delivered with each spray
- calculate the number of sprays needed to dispense the volume of formulation in one dose (assume specific gravity = 1)
Keep in mind that the patient’s manner of squeezing the spray bottle may be different. The pharmacist should have the patient calibrate the spray bottle to provide more accurate dosing.
How to Use Intranasal Drops
- Blow your nose gently to clear the nostrils.
 Wash your hands with soap and warm water.
Wash your hands with soap and warm water.- Lie down on a bed with your head tilted back and the neck supported (allow the head to hang over the edge of the bed or place a small pillow under your shoulders). Tilt your head back so that it is hanging lower than your shoulders. Note: If putting drops into the nose of a child, lie the child on his or her back over your lap. The head should be tilted back.
- Draw up a small amount of medication into the medicine dropper.
- Breathe through your mouth.
- Place the tip of the medicine dropper just inside your nostril (about 1/3 inch). Avoid touching the dropper against the nostril or anything else.
- Place the directed number of drops into your nostril.
- Repeat steps 3 – 7 for the other nostril if directed to do so.
- Remain lying down for about 5 minutes, so the medication has a chance to spread throughout your nasal passages.
- Replace the medicine dropper to its container and tightly close the bottle.
- Wash your hands.
Comments:
- Some of the solution may drain down into your mouth. If the taste is unpleasant, you may cough out the excess solution into a tissue.
- It may be much easier to have someone help you instill your nose drops.
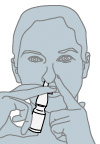 How to Use Intranasal Sprays or Pumps
How to Use Intranasal Sprays or Pumps
- Blow your nose gently to clear the nostrils.
- Wash your hands with soap and warm water.
- Hold your head in an upright position.
- Close one nostril with one finger.
- With the mouth closed, insert the tip of the spray or pump into the open nostril. Sniff in through the nostril while quickly and firmly squeezing the spray container or activating the pump.
- Hold your breath for a few seconds and then breathe out through your mouth.
- Repeat this procedure for the other nostril only if directed to do so.
- Rinse the spray or pump tip with hot water and replace the cap tightly on the container.
- Wash your hands.
Dihydroergotamine Nasal Spray: scenario and formulation record.