Gels
Upon completion of this exercise, you should be able to:
- Explain the general structure of a gel.
- Identify common gelling agents used in compounded formulations.
- Differentiate the properties of:
- water soluble gels, and
- alcohol soluble gels.
- Explain how a water soluble gel can create a modified release compounded formulation.
Gels are an excellent formulation for several routes of administration. They are useful as liquid formulations in oral, topical, vaginal, and rectal administration. Gels can be clear formulations when all of the particles completely dissolve in the dispersing medium. But this doesn’t occur in all gels, and some are, therefore, turbid. Clear gels are preferred by patients.
Gels are made using substances (called gelling agents) that undergo a high degree of cross-linking or association when hydrated and dispersed in the dispersing medium, or when dissolved in the dispersing medium. This cross-linking or association of the dispersed phase will alter the viscosity of the dispersing medium. The movement of the dispersing medium is restricted by the dispersed phase, and the viscosity is increased.
If the gel contains small discrete particles, the gel is called a two-phase system. If the gel does not appear to have discrete particles, it is called as a one-phase system. Two-phase systems are thixotropic; e.g., they are semisolid on standing but liquefy when shaken. If the particle size in a two-phase system is large, the gel is referred to as a magma.
Examples of two-phase systems include Aluminum Hydroxide Gel and Bentonite Magma. Single-phase systems contain linear or branched polymer macromolecules that dissolve in water and have no apparent boundary with the dispensing medium. These macromolecules are classed as natural polymers (i.e., tragacanth), semisynthetic cellulose derivatives (i.e., methylcellulose), or synthetic polymers (i.e., Carbomer polymers). Single-phase gels made from synthetic or natural macromolecules are called mucilages.
Common Gelling Agents
There are many gelling agents. Some of the common ones are acacia, alginic acid, bentonite, Carbopols (now known as carbomers), carboxymethyl cellulose, ethylcellulose, gelatin, hydroxyethyl cellulose, hydroxypropyl cellulose, magnesium aluminum silicate (Veegum), methylcellulose, poloxamers (Pluronics), polyvinyl alcohol, sodium alginate, tragacanth, and xanthan gum. Though each gelling agent has some unique properties, there are some generalizations that can be made:
- If the gelling agent is added to the dispersing medium in a haphazard manner, there is a tendency for the agent to “clump.” The outer molecules of the gelling agent contact the medium first and hydrate, forming a surface layer that is more difficult for the medium to penetrate. The clumps will ultimately hydrate, but it will take more time. A much more efficient manner is to sieve the agents onto the surface of the medium a little at a time as the medium is stirred. Using glycerin as a wetting agent will sometimes minimize clump formation.
- Some gelling agents are more soluble in cold water than in hot water. Methylcellulose and poloxamers have better solubility in cold water while bentonite, gelatin, and sodium carboxymethyl cellulose are more soluble in hot water. Carbomers, tragacanth, and alginic acid gels are made with tepid water.
- Some gelling agents (carbomers) require a “neutralizer” or a pH adjusting chemical to create the gel after the gelling agent has been wetted in the dispersing medium.
- Most gelling agents require 24 to 48 hours to completely hydrate and reach maximum viscosity and clarity.
- Gelling agents are used concentrations of 0.5% to 10%, depending on the agent.
- It is easier to add the active drug before the gel is formed if the drug doesn’t interfere with the gel formation.
- Only Carbopol 934P, methylcellulose, hydroxypropyl methylcellulose, and sodium carboxymethyl cellulose are recommended for oral administration.
Carbomer is a generic name for a family of polymers known as Carbopol. Carbopols were first used in the mid 1950s. As a group, they are dry powders, which have high bulk densities and form acidic aqueous solutions(pH around 3.0). They thicken at higher pHs (around 5 or 6). They will also swell in aqueous solution of that pH to as much as 1000 times their original volume. Their solutions range in viscosity from 0 to 80,000 centipoise (cps). Some examples of this group of gelling agents are:
| Polymer Name | Viscosity* | Properties |
|---|---|---|
| Carbopol 910 | 3,000 – 7,000 |
|
| Carbopol 934 | 30,500 – 39,400 |
|
| Carbopol 934P | 29,400 – 39,400 |
|
| Carbopol 940 | 40,000 – 60,000 |
|
| Carbopol 941 | 4,000 – 11,000 |
|
* 0.5% solution, pH 7.5
Carbomer polymers are best introduced into water by slowly sprinkling a sieved powder into the vortex created by rapid stirring. This should prevent clumping. Once all of the powder has been added, the stirring speed should be reduced to decrease the possibility of entrapping air bubbles in the formulation.
As mentioned, when the carbomer is dispersed, the solution will have a low pH. A neutralizer is added to increase the pH and cause the dispersion to thicken and gel. Some neutralizing agents are sodium hydroxide, potassium hydroxide, and triethanolamine. If inorganic bases are used to neutralize the solution, a stable water soluble gel is formed. If triethanolamine is used, the gel can tolerate high alcohol concentrations. The viscosity of the gel can be further manipulated by propylene glycol and glycerin (to increase viscosity) or by adding electrolytes (to decrease viscosity).
The cellulose derivatives (methylcellulose, hydroxyethyl cellulose, hydroxypropyl cellulose, hydroxypropyl methylcellulose, and carboxymethyl cellulose) are commonly used. There are some commonalties in these compounds, and each one has their unique properties.
Common Traits
- The most difficult task is to introduce the compounds into solvents without or clumping. There are some compounding techniques that can be used to minimize this problem:
- sift the powders into the vortex of the rapidly stirring solvent
- levigate the powder with a water miscible nonsolvent such as absolute alcohol or propylene glycol
- use a blender to homogeneously mix the powder and solvent
- All of the derivatives except carboxymethylcellulose (CMC) maintain their viscosity over a wide pH range (3-11). CMC has its ideal viscosity at pH 7-9; the viscosity dramatically decreases below pH 4 and above pH 10.
- The compounds’ ability to hydrate is reduced by the addition of salts. Some derivatives are more sensitive to divalent and trivalent inorganic salts.
Unique Traits
methylcellulose 1500 cps
- Makes thinner gels with high tolerance for added drugs and salts
- Compatible with water, alcohol (70%), and propylene glycol (50%)
- Hydrates and swells in hot water. The powder is dispersed with high shear in about 1/3 of the required amount of water at 80°C to 90°C. Once it is dispersed, the rest of the water (as cold water or ice water) is added with moderate stirring.
- Maximum clarity, hydration, and viscosity will be obtained if the gel is cooled to 0-10°C for about a hour.
hydroxyethylcellulose
- Makes thinner gels
- Compatible with water and alcohol (30%)
- Hydrates and swells in cool water (about 8 – 12 hours)
- Forms an occlusive dressing when lightly applied to the skin and allowed to dry
hydroxypropylcellulose
- Makes thinner gels with high tolerance for added drugs and salts
- Compatible with alcohols and glycols
- Hydrates and swells in water or hydroalcoholic solution. The powder is sprinkled in portions into water or hydroalcoholic solution without stirring and allowed to thoroughly wet. After all of the powder is added and hydrated (about 8 – 12 hours), the formulation can be stirred or shaken.
- Good gelling agent if 15% or more of an organic solvent is needed to dissolve the active drug.
hydroxypropylmethylcellulose
- Makes thicker gels but lower tolerance for positively charged ions
- Compatible with water, alcohol (80%)
- Disperse in cool water
- Good gelling agent for time released formulations
carboxymethylcellulose
- Generally used as the sodium salt
- Makes thicker gels but less tolerance than hydroxypropylmethylcellulose. Maximum stability at pH 7 – 9
- Compatible with water and alcohol
- Disperse in cold water to hydrate and swell, and then heat to about 60°C. Maximum gelling in 1 – 2 hours.
Poloxamer (Pluronics) are copolymers of polyoxyethylene and polyoxypropylene. They will form thermoreversible gels in concentration ranging from 15% to 50%. This means they are liquids at cool (refrigerator) temperature, but are gels at room or body temperature. Poloxamer copolymers are white, waxy granules that form clear liquids when dispersed in cold water or cooled to 0-10°C overnight.
Pluronic F-127 is often combined with a lecithin and isopropyl palmitate solution to make what is called a “PLO gel.” This is a slight misnomer, since the final product is actually an emulsion. The confusion comes from using a gel as one of the ingredients for the emulsion.
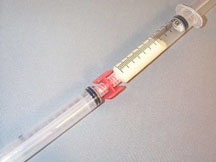 “PLO gel” is made by combining a Pluronic F-127 gel and a lecithin/isopropyl palmitate syrup. The two components are made and stored separately. When it is time to compound a formulation, water soluble drugs are dissolved in the Pluronic gel or oil soluble drugs are dissolved in the lecithin syrup. If a small quantity of formulation is to be made, each of the components can be put into a syringe and the two syringes are connected by a adapter. The mixture is forced between the two syringes and the shear caused by the passing the mixture through the adapter will create the “PLO gel.”
“PLO gel” is made by combining a Pluronic F-127 gel and a lecithin/isopropyl palmitate syrup. The two components are made and stored separately. When it is time to compound a formulation, water soluble drugs are dissolved in the Pluronic gel or oil soluble drugs are dissolved in the lecithin syrup. If a small quantity of formulation is to be made, each of the components can be put into a syringe and the two syringes are connected by a adapter. The mixture is forced between the two syringes and the shear caused by the passing the mixture through the adapter will create the “PLO gel.”
Lidocaine Gel: scenario and formulation record.